Once again, folks, please don’t try this at home.
I’ve read that certain household cleaners are very bad for copper, but being me, I wanted to see for myself. I decided to apply some forbidden cleaners to copper: hydrogen peroxide, bleach, and muriatic acid. (Okay, muriatic acid is not a household cleaner, really, but I happen to have some for the swimming pool and I wanted to see what it does.)
My first objective was to see what each chemical did to the copper.
- I expect the hydrogen peroxide, H2O2, to react with the copper to produce copper oxides, CuO and Cu2O. I’m using the 3% concentration stuff you buy at the pharmacy so it’s not too strong.
- I expect the chlorine bleach — sodium hypochlorite, NaClO, with caustic soda, NaOH — to react with copper to produce cupric chloride, CuCl2, and possibly cupric hydroxide, Cu(OH)2. I’m using concentrated Clorox bleach at 8.25% concentration.
- Muriatic acid is a dilute form of hydrochloric acid, HCl. I’m using a pool chemical that is labeled at 31.45%. It’s pretty nasty stuff, but the Internet tells me that HCl will not react with copper at all. We’ll see.
I just did a whole post on all the various copper compounds that can form with photos of their colors. My hope is that I can identify the products of each reaction by the colors I see.
My second objective was to see how serious the effects were. Did the reactions produce surface tarnish that I could subsequently erase? Or would there be permanent effects, like etching or pitting?
Here’s how I ran the experiment.
- I cleaned the base of a copper pot with copper cleaner to remove tarnish and washed it with soap and water to remove the cleaner.
- I placed hydrogen peroxide, bleach, and muriatic acid on sections of the base of a copper pot. I did not allow the acid and bleach to come into contact.
- I let them sit for an hour, taking photos every ten minutes or so.
- I rinsed and washed the areas with soap and water.
- I cleaned the areas with copper cleaner to remove surface compounds.
Starting point
As above, I cleaned tarnish from the copper and then cleaned chemical residue from the surface. Here’s the cleaned and marked pot. (The black spots are just carbonized grease — this is not one of my better pots.)

Here we go!
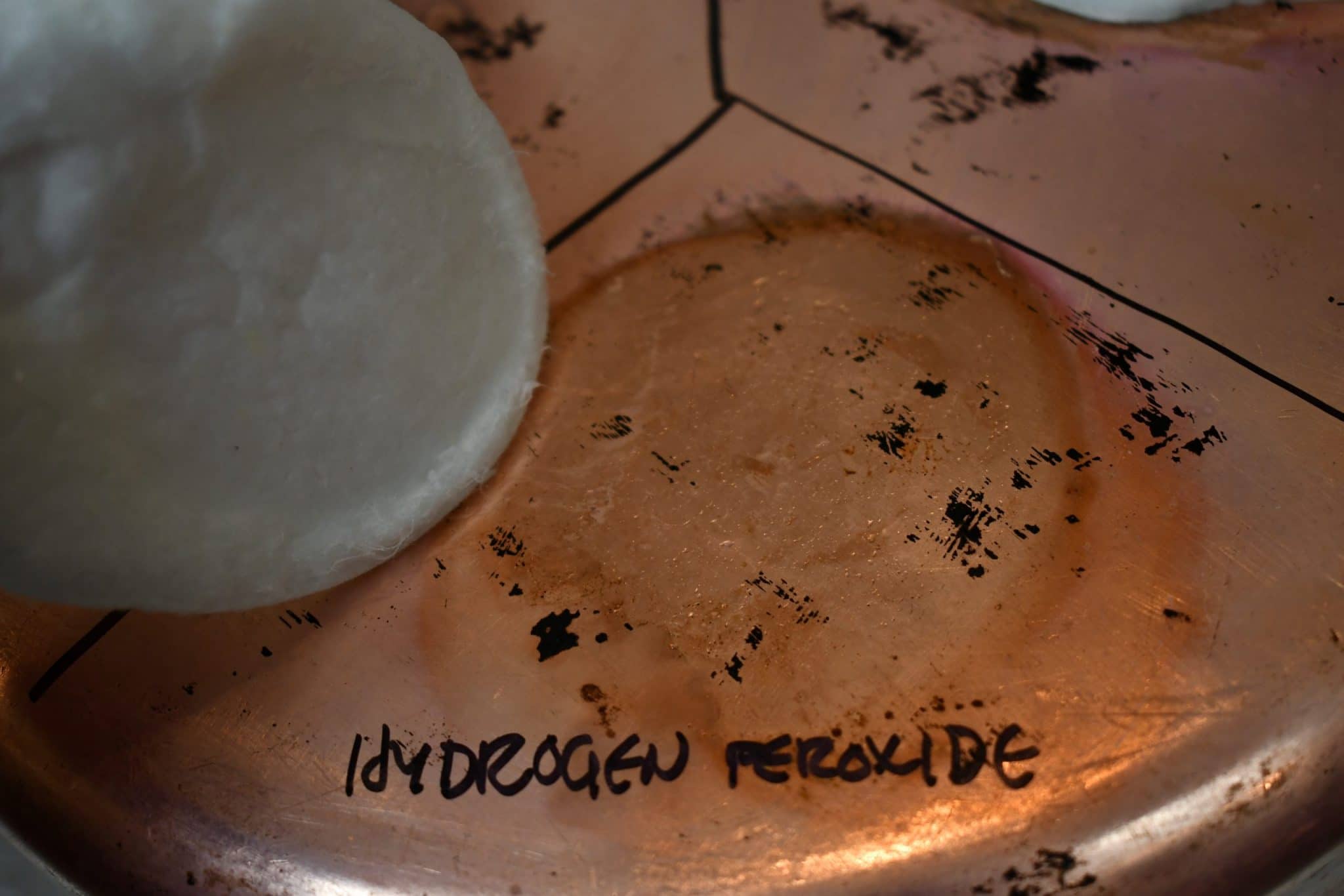
Hydrogen peroxide (H2O2, 3%)







Chlorine bleach (NaClO, 8.25%)
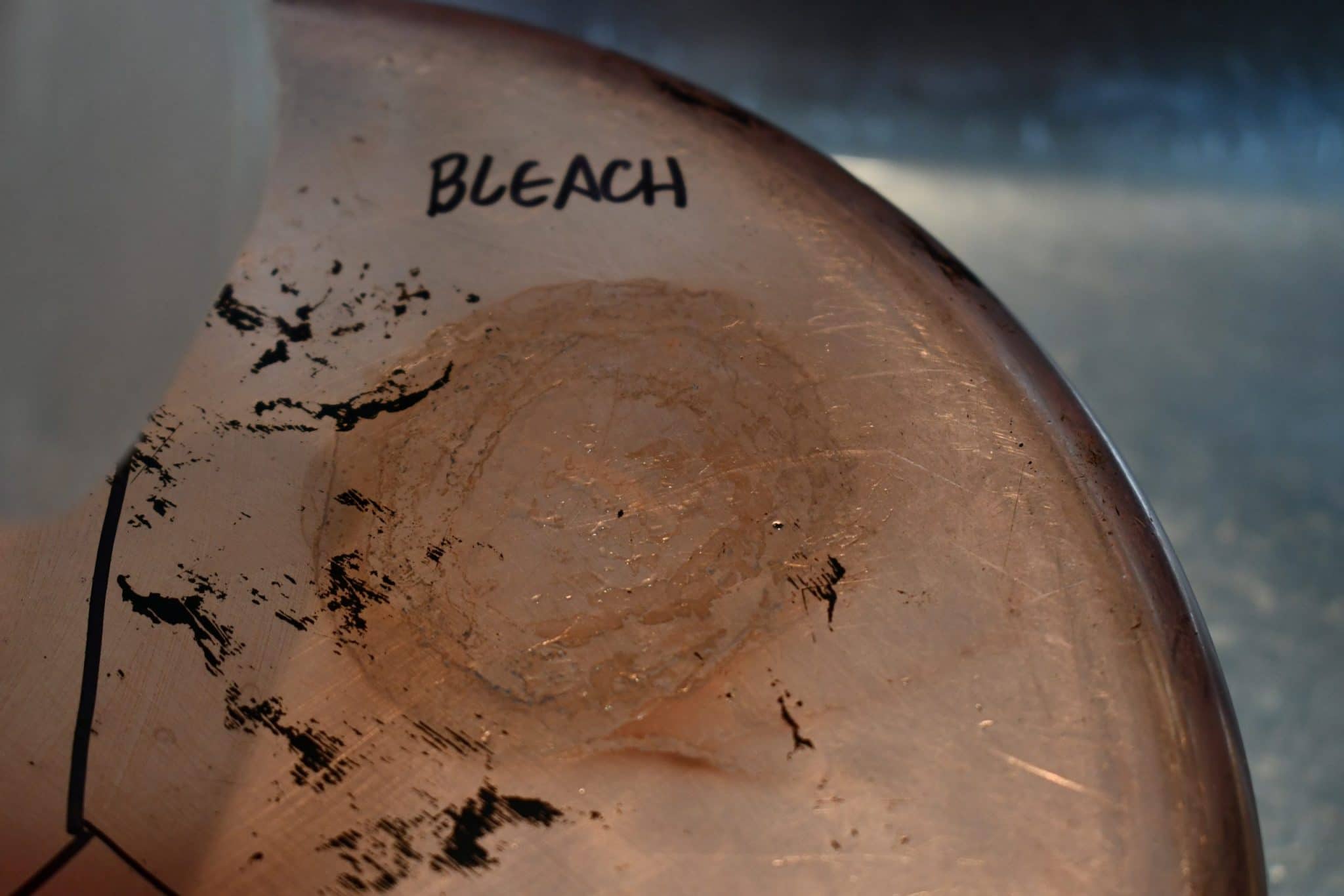







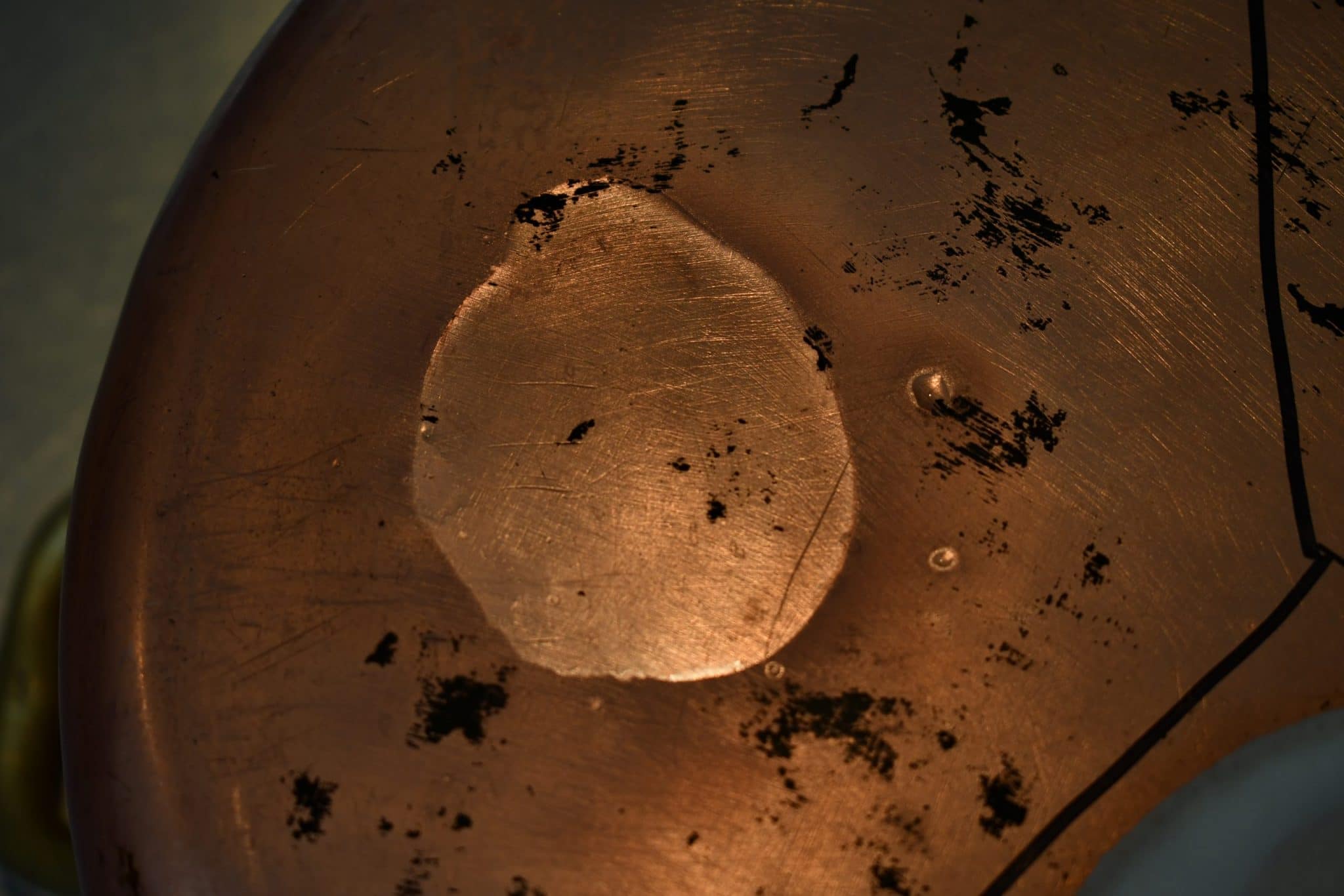
Muriatic acid (HCl, 31.45%)







End states
Hydrogen peroxide
 |
The hydrogen peroxide results are about what I expected. I predicted that the H2O2 would produce CuO (a red compound) and Cu2O (a black compound), and the ring around the reaction area is reddish and brownish. |
Chlorine bleach
 |
I expected that the chlorine in the beach would produce copper chloride, and I think that is indeed what is showing up. Copper(II) chloride is a blue-green compound, but the bright green had me stumped until I did some more research (see below). |
Muriatic acid
 |
The Internet reassures me that copper and muriatic acid don’t react, so I’m not sure what’s going on here. The acid is certainly etching the heck out of the copper. I also saw some bubbles forming along the edge of the puddle; I’d cleaned the tarnish off the copper, but I suspect copper oxides were spontaneously forming and then reacting with the acid. But I still don’t know what was going on under the puddle. |
What’s the bright green stuff?
I noticed a bright acid-green color around the perimeter of the bleach spot, and it didn’t match up with the color of copper(II) chloride, which is blue-green.
I saw the same color again when I neutralized the muriatic acid puddle with baking soda (NaHCO3). The spot fizzled and bubbled as the acid and base worked on each other, and it turned blue-green with copper(II) chloride) but also, again, bright yellow-green. What could it be?



I did some research and what I think is happening is that the chemical reactions are producing water (H2O) in addition to the copper(II) chloride, and the water enables a second chemical reaction to occur. The copper(II) chloride dissolves into the water and if there are more chlorine atoms floating around, they join up too and create CuCl4, an aqueous variant of copper(II) chloride that is green, not blue-green. The more chlorine, the more CuCl4, and the greener the liquid gets.
What I think is happening is that the bleach reaction and the muriatic acid + baking soda reaction both produced H2O. Some of the copper(II) chloride dissolved into the water, and then sucked up more chlorine and shifted to green.
Soap and water wash
After an hour I rinsed everything off and washed the surface with soap and water. Some of the surface stuff wiped right off — here’s how it looked.

Cleaning with copper cleaner
But there was also true tarnish — copper compounds on the surface that wouldn’t wipe or rinse off. I used copper cleaner to remove them.


Final results

Overall, each of the chemicals reacted with the copper, which explains the prohibitions against using them. A good copper cleaner reacts with what’s on top of the copper and not the copper itself.
The hydrogen peroxide produced tarnish but not lasting damage. The reaction I saw produced brown copper oxides but aside from the copper atoms stolen to form these there was no lasting damage.
The bleach area produced blue-green tarnish and also light etching. This light etching is evidence of some loss of copper but is not permanent, as it can be erased with an abrasive polish.
But the muriatic acid area is severely etched. It is rough to the touch and unlike anything I’ve seen on a copper pot. I’m honestly bewildered at what happened chemically, because multiple sources on the Internet reassure me that HCl on its own doesn’t react with copper. Acid etching of copper with HCl is a thing but requires hydrogen peroxide to activate the reaction. I was very careful not to let any of the test chemicals touch each other and I have a hard time imagining that the hydrogen peroxide could have snuck over into the acid.
I’m going to keep researching this. The etched spot doesn’t bother me — this was a sacrificial pot — but not knowing the chemistry bugs me. Any ideas?


I’m looking for advice. I have an old house with copper pipe, and I’ve recently had a pipe leak in a gray water exit pipe that was severely corroded. I’m trying to figure out what household cleaners may have contributed to this problem so I can eliminate them from my home. I’m not finding much information. Do you think the bleach such as in the washer or even as a household cleaner would be corrosive in the long-term?
Hi Stacie! I am so sorry to hear of your problems with your copper pipes. I have no experience with copper pipes for wastewater and therefore I’m not aware of specific product warnings. This page from Engineering Toolbox lists copper’s resistance to various chemical substances:
https://www.engineeringtoolbox.com/copper-tubes-corrosion-resistance-chemicals-d_2125.html
I hope that list gives you a place to start, but bear in mind that household products contain relatively low concentrations of chemicals in order to make them safe for consumer use. A plumber would be the best resource for experience-based advice on what products to avoid.
Hi Stacie,
I’ve had issues with pinhole leaks in copper hot water lines at joints as well as one cold water joint leak. I’ve heard and read of many potential causes, such that I’ll probably never be able to determine cause by process of elimination. If the pipe was outside the house, it could even be external environmental chemistry and not a household product (I have verdigris on an exterior copper overflow pipe from a boiler). I doubt that velocity or pressure are your problem on a drain. The best advice that I could give is to cut off as much copper pipe as you can, buy some SharkBite fittings and PEX pipe, and replace the copper line. If you have a plumber conduct a repair, ask for the pipe to be replaced with PEX or PVC.