I thought I’d start with the chemistry of copper tarnish.

Tarnish is not one thing but rather a buildup of multiple copper compounds over time that create a layer of patina on the surface of your pot. Even as your copper pot is sitting quietly on your shelf awaiting its chance to help you with your next meal, its surface is alive with a slow and steady churn of chemical reactions. These reactions don’t hurt the copper — on the contrary, they’re a self-protective mechanism of sorts, forming a tough outer skin that shields the pure copper within.
The compounds that we call copper tarnish are collectively called copper salts. They form when copper oxidizes from contact with airborne oxygen, carbon dioxide, sulfur dioxide, and hydrogen sulfide, as well as the chlorine and various carbonates in tap water. Which compounds you’ll see on your copper depends on what’s in your environment and how long they’re left in place. If you live in a humid place, the moisture on your copper’s surface facilitates the oxidation process by helping to capture molecules that float by.
Copper salts
There are more than I list here but I am trying to focus on the simpler ones most likely to appear on your copper pots as you use them.
Oxygen and copper can form copper oxides.
 |
Copper(I) oxide (Cu2O, cuprous oxide) shades from red to yellow. It’s usually the first tarnish to show up. It’s yellow at higher temperature, and it’s what turns your copper brassy yellow when you cook on high heat. |
 |
Copper(II) oxide (CuO, cupric oxide) is black. This compound requires some heat energy to form, so it starts showing up when you use your copper pot for cooking. |
Sulfur and copper can form copper sulfides.
 |
Copper(I) sulfide (Cu2S) is dark gray. It occurs in nature as the mineral ore chalcocite, which is the primary source for mined copper. |
 |
Copper monosulfide (CuS) is dark indigo blue. Its mineral form, called covellite, is much more rare than chalcocite. |
Sulfur, copper, hydrogen, and oxygen can form copper sulfates.
 |
Copper(II) sulfate (CuSO4(H2O)x, cupric sulfate) changes color with moisture. It’s white when dry and blue when hydrated. |
Copper and chlorine can form copper chlorides.
 |
Copper(I) chloride (CuCl, cuprous chloride) is white. Its industrial use is primarily to make fungicides. | |
 |
 |
Copper(II) chloride (CuCl2, cupric chloride) is another copper salt that changes color. It is brown when dry and bright green when hydrated. |
Copper and hydrogen can form copper hydroxides.
 |
Copper(II) hydroxide (Cu(OH)2) was one of the first copper compounds made by alchemists and was used as a blue pigment for paint. |
If left in place, these compounds will continue to react and recombine until they turn into basic copper carbonate. Its building blocks — copper (Cu), carbonate (CO3), and hydroxide (OH) — can combine in two formulas to produce two distinct compounds.
 |
Cu2CO3(OH)2 is the green mineral malachite.
|
 |
Cu3(CO3)2(OH)2 is the blue mineral azurite. |
Verdigris
 The word verdigris is thrown around to describe any blue-green copper tarnish, but if you’re going to cook with copper you need to understand the different uses of the term.
The word verdigris is thrown around to describe any blue-green copper tarnish, but if you’re going to cook with copper you need to understand the different uses of the term.
Verdigris as a color refers casually to any of the several blue-green copper compounds that can form on the surface copper.
Verdigris as a patina refers to the uniform blue-green appearance of a copper surface that has weathered naturally. The patina is likely composed of several copper compounds but is primarily basic copper carbonate or sulfate that has formed over a period of years. The best-known example is the resilient patina on the Statue of Liberty that has protected the underlying copper for more than a century.
But verdigris as a chemical refers to a specific blue-green copper salt, copper(II) acetate, that is mildly toxic. A bit of verdigris in your food won’t kill you but it will make you and your guests sick to your stomach. It will look like a bit of bright blue-green crust and if you see it on any copper used for eating or drinking you should clean it off immediately.
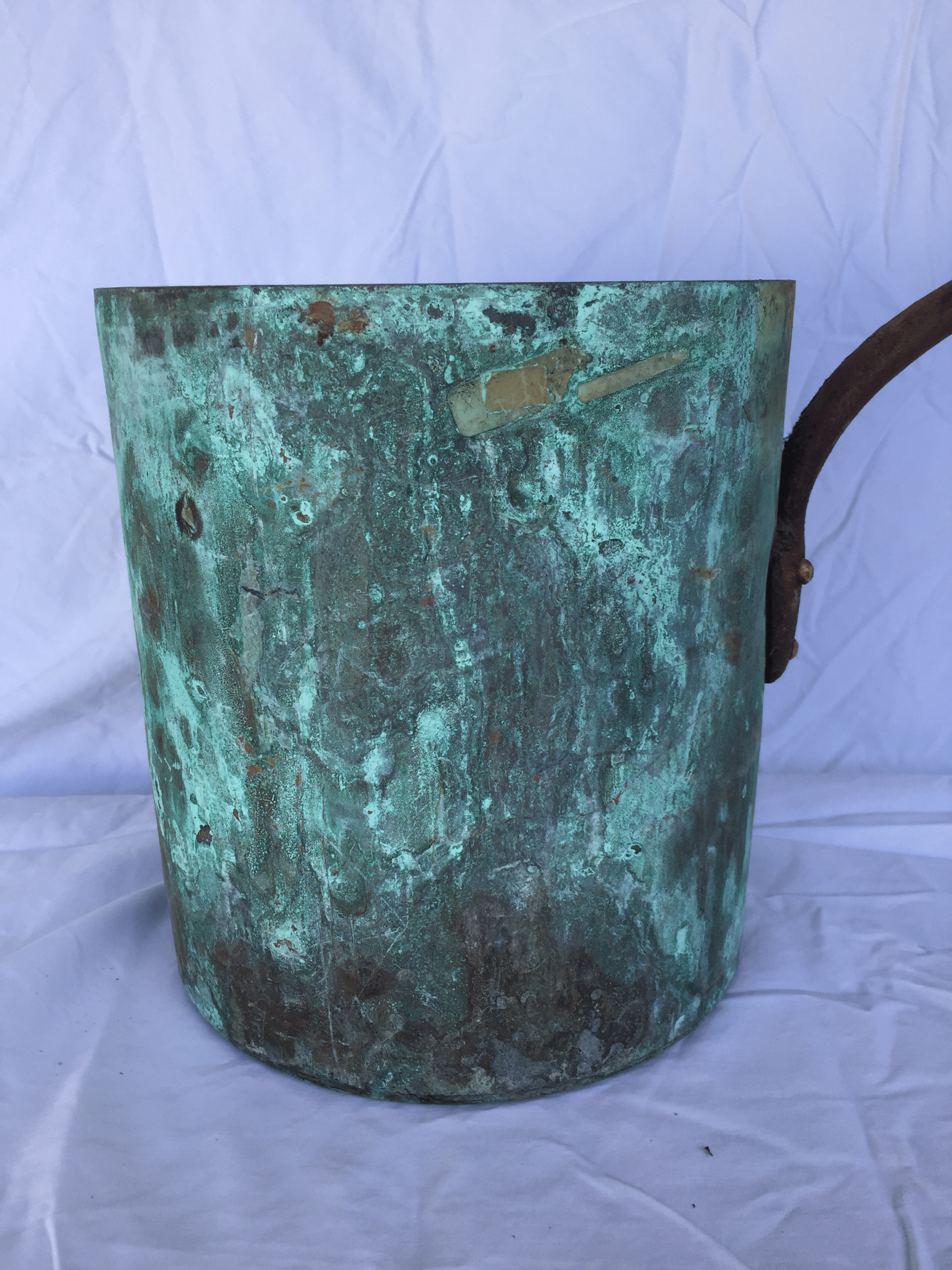

 |
Copper(II) acetate (Cu(CH3COO)2, cupric acetate) is toxic verdigris that should be removed immediately from copper used for food or drink. |
True verdigris can be formed fairly quickly and easily by combining vinegar — acetic acid — with copper oxide, copper hydroxide, or basic copper carbonate. When verdigris was used to make pigment for paint, people would submerge copper in vinegar or wine for several days and then scrape off the verdigris and grind it into pigment. Copper jewelry makers and other artisans who want to produce verdigris as a patina on copper use heat and concentrated acetic acid to speed up the process.
Again, the danger is that verdigris forms on a cooking surface and dissolves into food or beverages. Verdigris on the outside of a copper pan is not likely to do this. However, it’s worth developing a sharp eye for patches of bright blue-green and removing them.
Good news, bad news
The good news is that tarnish doesn’t hurt your copper pot. (Even verdigris, which is toxic to us, doesn’t hurt copper.) Some metal oxidation is destructive — for example, when iron oxidizes into rust, the resulting ferrous oxide detaches and flakes off, exposing the underlying iron to further oxidation and flaking. But tarnish doesn’t do that. Instead of flaking off, the copper compounds stick to the surface, forming a physical barrier between the air and the underlying un-tarnished copper. (By the same token, that tight bond also makes tarnish difficult to remove.)
The bad news is that the layer of tarnished copper interferes with light, so your copper loses its shine. As more and more of the zillions of copper atoms on the surface oxidize, they stop reflecting light and instead refract it into browns and yellows. Over time, and accelerated by the heat of cooking, the surface of the copper turns dull brown. Cleaning tarnish off your copper means stripping away this outer layer of oxidized copper and, yes, exposing the underlying layer to another round of oxidation.
If you want to prevent the oxidation entirely, you’re going to have to keep the copper from coming into contact with air. One way to do this is to cover the surface with a thin layer of oil, which is what copper polish does — it comes in a greasy form in a tube. Even when you wipe the polish away, it leaves a thin layer of grease behind that seals off the copper from the air and keeps the underlying copper atoms bright and un-oxidized.
Conclusion
From a distance, a well-used copper pot looks like a uniform brown color. But if you look more closely at your tarnish, you will start to see some more colors embedded in it. The rainbow is a mosaic of copper compounds: red, yellow, blue, green, brown, gray, and black. Large patches of one compound create a single sheen, while in other areas, multiple compounds may overlay each other to create intermediate shades of purple and orange. It’s beautiful and chaotic.
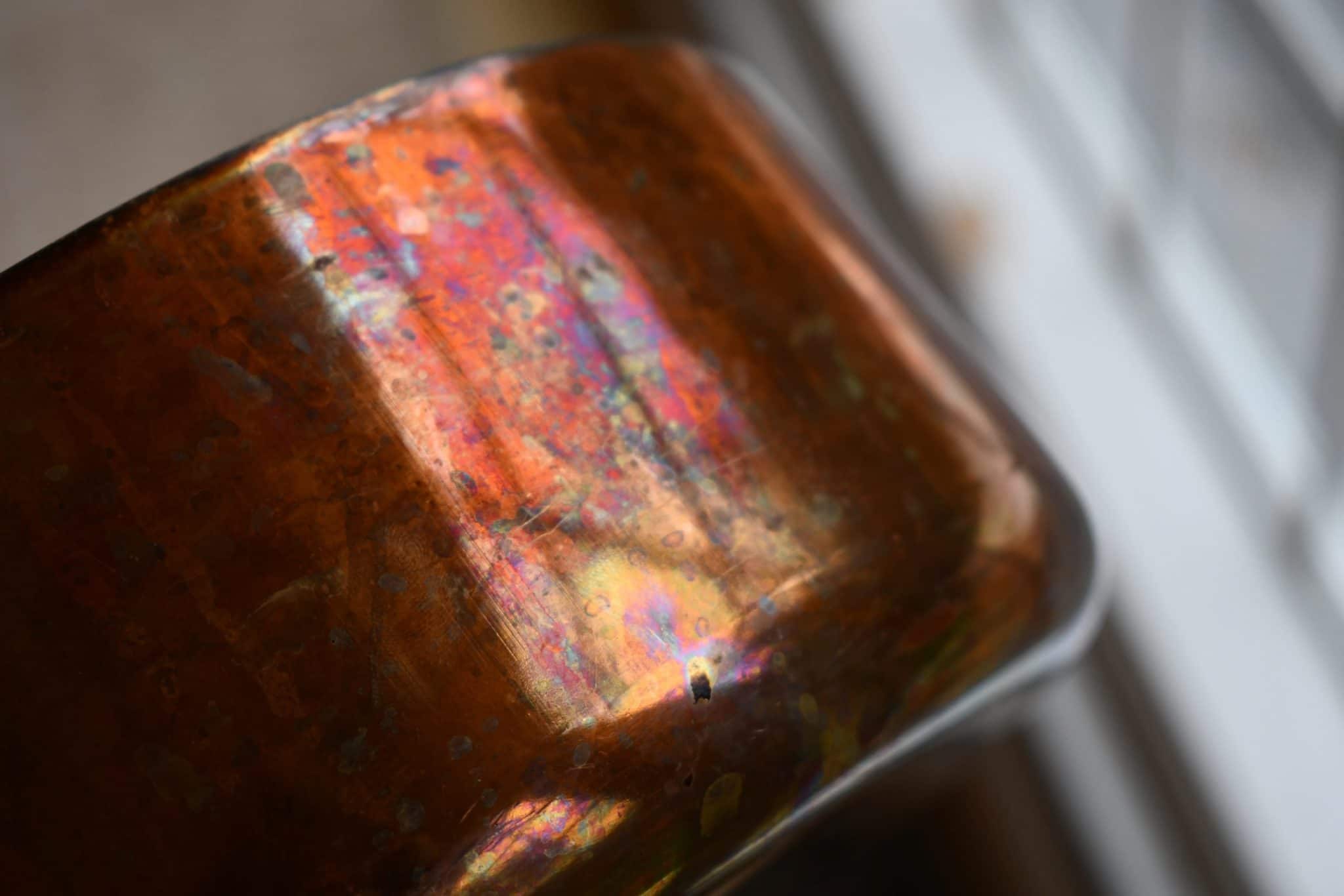
In Part 2, I discuss how to remove tarnish… that is, if you actually want to remove it!



